Artificial hair curling actually goes back to the time of Cleopatra. At that time, people plastered basic (alkali) mud on their hair, coiled it around a stick, dried it, and washed it to make wavy hair. It is not quite like how they make permanent wave these days, but the principle is almost the same. How?
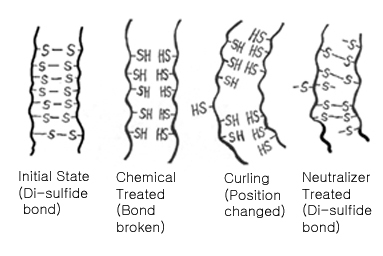
To understand this process, you must first know what a hair is made up of. Hair mostly consists of keratin, a double stranded protein with many cysteine molecules cross linked by disulfide bonds, like in the rungs of a ladder. (Refer to the illustration below.) When these cysteine molecules are linked in a straight line, the hair stays straight.
In the permanent wave process, a basic reducing substance (usually ammonium thioglycolate) is added to break some of the sulfur-sulfur links. Then, the hair is put on rollers or curlers. Since the bonds are no longer tightly cross-linked to each other, they can shift positions in relation to each other. After a while, an oxidizing agent (usually a dilute solution of hydrogen peroxide), also called a neutralizer, is added to reform the disulfide bonds in their new positions, which makes the hair hold its new, curled shape.
An interesting thing is that natural red hair holds a better wave pattern for a longer period of time than others because red hair contains about 8 percent sulfur compared to the average persons hair containing about 4 percent sulfur. Red hair will be harder to perm because there are more sulfur-sulfur bonds to break down, but once the desired curl pattern is achieved there are many more bonds to make the wave pattern last stronger and longer.
Different ethnic backgrounds and genetic makeups also suggest that the sulfur content of cysteine varies greatly for each individual person. So, for minimum damage to your hair, a chemical analysis of the hair is needed to choose the right type of perming solution and the right amount of time for the chemical to break the disulfide bonds.
The only sad thing is that the permanent wave will eventually grow out. The new disulfide bond positions will be continuously influenced by shampooing (and other chemical reactions), and of course, the new hair growth is not treated.